This science study guide will help you to convert the units of base physical quantities from one system to another with ease. At the end is a list of commonly used units along with their conversions and some exercises to absorb the concepts learned.
Different Systems of Units
Have you ever been perplexed by the units used for measurement in your science homework questions? As science developed and new quantities were introduced, different countries have adopted different standards for measurement. This has resulted in the need for carrying out varied unit conversions. In this article, the words “metre” and “meter” are interchangeable.
- The Standard International(SI) system of units or the metre-kilogram-second system of units. This is the most commonly used and internationally accepted system of units. It defines units of 9 basic physical quantities and the units of the derived quantities can be obtained from these base units.
Also defined are certain prefixes that can be used with the main units.For example, using the prefix “kilo” before metre makes a kilometre. A kilo means 103, so a kilometre = 103 metre.
2. Imperial Units or the foot-pound-metre system of units. This is the British system of units and is still used in a lot of books, though the SI system is preferred.
3. Industrial units which are defined taking special considerations.
4. Also, there is the metric system and c-g-s system.
In spite of the multitude of available units, you need not worry because once you understand the basic fundamentals, you can crack them all.
1. The Conversion factor
If you know the conversion factor, you can easily convert one quantity to another. This is a very important concept, make sure you understand this well. Let us take an example.
Say we are measuring the length of a body. The question quotes the value as i inches. We have to convert this to metres. Let’s say the value comes out to be m metres. We are required to calculate m.
Definition: The conversion factor is defined as the number by which a value that is expressed in one set of units must be multiplied in order to convert it into another set of units.
So, in our example, we need to find out c, the conversion factor, such that i multiplied by c gives m.Once you know this conversion factor, c, you can convert any measurement in inch to metre.
i X c = m
Also, in order to do the reverse of this conversion, that is from metre to inch, you only need to reverse the conversion factor. The reverse of the conversion factor is its reciprocal, 1/c. So, m multiplied by 1/c gives i.
m X 1/c = i
This has been obtained from the first equation itself.
Calculating the Conversion Factor
The conversion factor can be calculated by the unitary method.
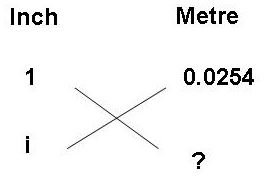
Make a table as shown above in the diagram. Put a question mark at the quantity to be calculated. The unknown quantity can be calculated by multiplying the diagonal quantity and dividing this by the only other known quantity in the table.
So here, the unknown quantity m = i X 0.0254/1
The conversion factor here is 0.0254/1 = 0.0254
The next thing that we have to tackle is calculating how many inches make a metre, that is, how was the quantity 0.0254 obtained?
Obtaining a Relation Between the Units
The question now left to be answered that would give us the complete solution is a relation between the two units, of the form:
1 inch = x metres
Since we are using the unitary method, we need not have the conversion of “1” inch into metres, we may be given any conversion of inch into metres. Here comes the use of predefined relations between units.
1 inch = 2.54 cm is a well known conversion, in most circumstances you will be expected to know this or this may be given in the question itself. I have given at the end of this article, a list of some basic units which are very commonly used, make sure you go through them.
Also, 100 cm = 1 metre. You should definitely know this one. Go through the link of prefixes in the first section of the article as well as the ones given in the list at the end.
So, reverting back to the unitary method,
1 metre 100 cm
x metre 2.54 cm
x metre = 2.54 cm X 1 metre / 100 cm
Note that the cm gets canceled and just metre remains on the R.H.S.
2.54/100 metre = 2.54 cm = 1 inch.
So we have,
1 inch = 0.0254 m
Likewise, all base unit conversions can be done by using the unitary method, provided you know the relations between the units.
A List of Relations Between Units
Here are some of the commonly used units for length, mass, time & temperature. Use them to do the exercises at the end so that you remember the conversions.
Length
S.I. system : m (metre)
Imperial system : ft (foot)
0.01 m = 1 cm = 2.54 inches = 1/12 feet = 0.083 feet
common prefixes used in SI : nano(10-9), micro or μ(10-6), milli(10-3), centi(10-2), deci(10-1), kilo(103), mega(106), giga(109)
For example, 1 nanometre = 10-9 metre
Mass
S.I. system : Kg (kilogram)
Imperial system : lb(pound)
1 kg = 2.2 pounds
Commonly used prefixes : 1 kg = 1000 g(grams)
Other units : 1 tonne = 1000 Kg
Time
S.I. system : s(second)
Imperial system : s(second)
Commonly used prefixes : micro(10-6), nano(10-9)
Other units :
1 minute = 60 s
1 hour = 60 minutes
1 day = 24 hours
Temperature
S.I. System : K (Kelvin)
Imperial System : oF (degrees Fahrenheit)
Other units : oC (degrees Celsius)
Note: The different units of most other quantities differ because they have different step sizes. However in case of temperature, the “0” has also been defined differently in each case. That is why we cannot use the unitary method here. You must use the conversions given below for these conversions.
Temperature in oC = Temperature in K - 273 K
(Temperature in o_F_ - 32) * 5 / 9 = Temperature in oC
Exercises
Take a pen and paper and carry out these conversions. Avoid a calculator and try not to look at the conversions given above, instead try recalling.
- 1 pound = _____ grams
- 273 K = ______ oF
- 1 minute = ______ nanoseconds 1 foot = _____ metres
This post is part of the series: Units & Dimensions Study Guide for High School Physics
A resourceful set of articles on units that will come in handy in physics class, as well as using units and dimensions to verify or derive formulae!