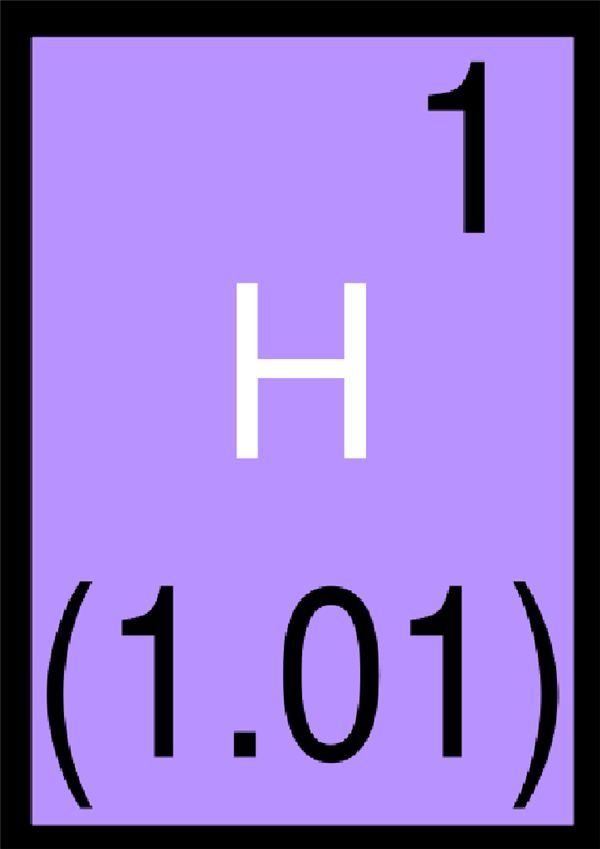
If you are trying to learn the elemental symbols in chemistry, flash cards are proven to help retain the knowledge you need. In this slideshow, you will see the symbol and be given the answer on the next page. Bookmark this guide to help you study for your next chemistry test!
What Element Does This Symbol Stand For?
Hydrogen
The correct answer is “Hydrogen”.
Did you know…
Hydrogen is found in group 1 of the alkaline elements. The metal is found in a gaseous state, with a boiling point of –252.87 degrees Celsius and a melting point of –259.34 Celsius. The electron arrangement is 1s1.
T. von Hehenheim found hydrogen. Hydrogen was used in filling balloons and airships, although it was found to be dangerous due to its high explosive nature. The Hindenburg is one example of how explosive hydrogen can be in terms of filling inflatable products. Helium is used for many items now in place of hydrogen.
Hydrogen fuels the stars. The high temperatures of stars are created by hydrogen atoms that hit into one another and join together to produce helium atoms. The reactions give out large amounts of energy such as heat and light. Hydrogen atoms are believed to be the first atoms to create forms in the Universe.
Space shuttles also use hydrogen in liquid form as fuel due to the amount of power it creates, while weighing hardly anything. Like other forms of fuel, it takes oxygen to burn. The shuttle will contain a tank of liquid hydrogen and a tank of liquid oxygen. The two liquids spray out together into the engines. As hydrogen explodes, the steam shoots out of the nozzles, producing the thrust the shuttle travels by.
What Element Does This Symbol Stand For?
Helium
The correct answer is “Helium”.
Did you know…
Helium was first discovered during a solar eclipse in 1868, when J.N. Lockyer and E. Frankland noticed a brighter yellow emission line coming from the sun. This was not corresponding to any known element and was named by the two witnesses. “Helium” comes from the Greek word, “helios” which means “sun”.
Deep-sea divers and caisson workers use helium in decompression chambers because it is less soluble in blood than nitrogen. Helium reduces the risk of the caisson disease known as “bends”. Used in arc welding and in growing crystals made of silicon and germanium for semiconductors, it has also been used in refining titanium and zirconium metals as well. Pressurizing fuel tanks of rockets that are liquid-fueled, liquid helium is a key element in low temperature applications of various types.
Helium-4 forms two different types of liquids. When cooled below 4.22K, it condenses into liquid helium-1. Helium-1 reacts as an ordinary liquid. When liquid helium-1 is cooled to about 2.18K, liquid helium-II is created. This chemical has numerous unusual properties. It is also known as a superfluid because of the extremely low viscosity levels, high heat conductivity and it expands when cooled. It can never be contained in an open container, since a thin film moves up the side, onto the edge and down the outside. Low-temperature physics studies these phenomena.
Which Element Does This Symbol Stand For?
Lithium
The correct answer is “Lithium”.
Did you know…
Lithium is a metallic chemical element which is number 3 on the elemental chart. It is a soft metal, silver-white in color. Lithium is placed in group 1 of the periodic table, in the alkali metals group. With high heat, it is used in some cooling systems for nuclear reactors. It is a very corrosive element and is prepared through electrolysis of fused lithium chloride. Lithium will not react with water as strongly as sodium. A brilliant white flame is seen when burned in combination with air. If added into small portions of magnesium, aluminum or lead-base alloys, it can be used to remove gas in steel, copper and iron refining.
Lithium can form numerous organic compounds, including lithium stearate, which is produced by cooking tallow or other animal fat, with lithium hydroxide. The lithium stearate is used to change oil into a lithium-base lubricating grease which is used extensively in the automotive industry. Special glasses and ceramic glazes are made from this same chemical compound. Lithium chloride and bromide help in the brazing and welding industry and used in air conditioning systems due to the moisture retaining property.
Electronic vacuum tubes use lithium to draw in small amounts of oxygen and nitrogen. Trace amounts of lithium and various compounds create a flame that is bright red which has some use in pyrotechnics. Lithium is a salt form that has recently come into use as one medical treatment for bipolar disorder. It is widely found in nature. It’s in plants, animals and even in the human body. The sun also houses lithium.
Mining ore allows for profit by extracting lithium from the hard ore. A few of the commercially key minerals are lepidolite, petalite, spodumene and amblygonite. Lithium is also located in brines such as the brines found in Searles Lake, California, and Great Salt Lake, utah.
What Does This Symbol Stand For?
Beryllium
The correct answer is “Beryllium”.
Did you know…
A strong, metallic element, with a placement of number 4, beryllium is an extremely light, silverish metal that contains a crystalline hexagonal structure. Known as an alkaline-earth metal, it is placed in group 2 of the periodic table.
With a high-melting type of formation, it is low in density and has found a place in the aerospace industry. It is like aluminum in that it is resistant to air oxidation even when under high heat. The components of beryllium have a sweet taste to them and are highly toxic. Beryllium is used in X- rays due to the ability to go beyond glass or other metals in this field.
Beryllium is soluble in hot nitric acid, diluted hydrochloric and sulfuric acids and sodium hydroxide. It is like aluminum and magnesium in many ways. It resembles the two chemically and it readily creates compounds with other elements. Beryllium can’t be found in nature. This chemical is resistant to the oxidation process when it is exposed to air. A protective oxide film prevents oxidizing, which is brought on naturally. The toxicity has unfortunately limited the use of beryllium as rocket fuel, although it does provide more heat on combustion for its weight than any other element. A desirable element due to a high melting point and because of transmitting X-rays better than gas or metals, it is used as a material for windows in high-intensity X-ray tubes. Beryllium is a great reflector and absorber of neutrons; shields and nuclear reactors also employ the chemical.
When beryllium is mixed with 2 to 3 percent copper, it produces a nonmagnetic alloy that is six times stronger than copper alone. Non-sparking tools are used in oil refineries and other places where sparks can cause a fire, making beryllium a perfect alloy. When it is mixed with other metals like aluminum or gold, it manufactures substances with an even higher melting point, with more hardness and strength and an even lower density than the metal it was alloyed with.
What Does This Symbol Stand For?
Boron
The correct answer is “Boron”.
Did you know…
Boron is a nonmetallic element that is dark brown to black. It is an amorphous powder or also is found as a hard, black to silver colored brittle, metal-like crystalline that is solid. Boron comes in either tetragonal or two rhombohedral shapes in the crystalline family. The chemistry of boron remarkably resembles the chemical make-up of silicon than those of the group it is found in – group 13. It is traditionally used in borax and boric acid.
Boron resembles the chemical make-up of silicon more than any of the other elements in Group 13 of the periodic table, in which it belongs as well. Depending on its form, decides the point of reactivity. In crystalline form, it is far less reactive than in amorphous form. The amorphous powder that is oxidized slowly at room temperature will ignite spontaneously at higher temperatures to form an oxide. The crystalline form is oxidized very slowly only, even at higher temperatures. Only a diamond is harder than boron carbide. It forms compounds with oxygen, hydrogen, the halogens, nitrogen, phosphorus and carbon. It is also an element that will form organic compounds. Boron absorbs neutrons and is used in shielding materials along with some control rods in nuclear reactors.
If boron is added to plastics, it can produce a material that is stronger than steel, yet remains lighter in weight than aluminum. You cannot find boron in nature. Deposits of borax, kermite, colemanite and other boron minerals lie in arid regions of the western United States. Tourmaline minerals contain boron.
Which Element Does This Symbol Stand For?
Carbon
The correct answer is “Carbon”.
Did you know…
As the sixth most common element, it is the element that is found in each and every living thing. Atoms made up of carbon are passed in from one living thing to another through the carbon cycle. Carbon dioxide is throughout our air and it also creates a host of other products, such as coal, crude oil, and natural gas. Carbon is rare in the purest form, however, it can be found in one of a number of different forms.
A diamond is the hardest known mineral, which houses atoms of carbon that are so tightly connected to each other that it creates a rigid grid known as a crystal lattice. The diamond is produced by compressing molten rock over millions of years.
Which Element Does This Symbol Represent?
Nitrogen
The correct answer is “Nitrogen”.
Did you know…
Proteins are vital to life and are partly much made of nitrogen. The nitrogen is recycled between plants and animals through air and ground. This recycling of nitrogen is called the “nitrogen cycle”. The air is 78% nitrogen, which is gas. Unreactive at regular temperatures, it is used in packaging to help keep the contents fresh. It is also used in the make-up of chemicals such as fertilizers and explosives.
Which Element Does This Symbol Represent?
Oxygen
The correct answer is “Oxygen”.
Did you know…
From 1742 – 1786, a Swedish chemist, Carl Scheele, discovered the chemical element of oxygen. He also found that air is not a single element. Oxygen makes up 21 % of our air, as a clear gas that has no odor. Water, minerals and almost every living thing have oxygen as an element in them. Oxygen molecules host double the oxygen atoms. The ozone layer is made up of a 3-atom form. The oxygen element is transferred from the air to plants, animals and humans and back again through the “oxygen cycle”.
Which Chemical Element Does This Symbol Represent?
Fluorine
The correct answer is “Fluorine”.
Did you know…
A gas from fluorine is manufactured through an electrolysis of hydrogen fluoride that hosts potassium hydrogen fluoride. Naturally, it is not found with other elements. Fluorine mixes spontaneously with the majority of other elements, with the exceptions of chlorine, nitrogen, oxygen, and inert gases such as helium or neon, to name a couple. It will react when incorporated with most inorganic and organic compounds.
What Element Does This Symbol Represent?
Neon
The correct answer is “Neon”.
Did you know…
Neon falls into the tenth place on the periodic table. It is a gas element, placed into group 18. A tiny portion of neon that is placed into an empty glass tube will emit a bright reddish-orange glow and give off electrical current. This rare gas is a part of the atmosphere in limited amounts. Collected as a byproduct when manufacturing liquid air, the most common use of neon is found in the neon signs in advertising. It is also used in automotive ignition timing lights, high-voltage warning indicators, in high-intensity beacons, and electron tubes and in Geiger counters.
What Element is This Symbol Used to Represent?
Sodium
The correct answer is “Sodium”.
Did you know….
The metallic element known as sodium is placed at number 11 in the periodic table. It is a softer, silver metal. Sodium reacts severely to other chemicals and is placed in the alkali metals group, number one, in the table. It is much like potassium, which it resembles in make-up, and will oxidize quickly when introduced to air. Water creates a violent reaction in sodium, freeing hydrogen and forms hydroxide. The hydrogen released may ignite.
Which Element is Represented Here?
Magnesium
The correct answer is “Magnesium”.
Did you know…
Magnesium is the 12th element listed in the periodic table. Sir Humphry Davy discovered magnesium in oxide form, although he wasn’t sure he isolated the metal. Magnesium was then isolated by A. A. B. Bussy in 1828, removing some of the chemical chloride. Magnesium is a silver, chemically active metal that has a hexagonal shape as of that found in a crystalline structure. One of the alkaline-earth metals, it is placed in group 2 of the table.
Which Element Does This Symbol Represent?
Aluminum
The correct answer is “Aluminum”.
Did you know…
Called “aluminium” in British countries and “aluminum” in the United States, this metallic chemical element sits at number 13 on the table. It is a silver metal with a cubic crystalline form. Group 13 of the periodic table is where aluminum resides. A conductor of heat and electricity, the pure metal is soft although it becomes hard when it is alloyed. It is known to be less conductive than that of copper wire, however, the aluminum wire is used in high-tension power transmission due to the lighter weight and the lesser cost. It is chemically reactive but resists corrosion through its oxide coating. Aluminum is the most commonly found metal in the earth’s crust.
Which Chemical Element Does This Symbol Represent?
Silicon
The correct answer is “Silicon”.
Did you know…
At periodic table position number 14, silicon is in group 14 of the periodic table too. Silicon boasts more metallic properties than does carbon but in various ways it resembles that of germanium. Silicon comes in two forms, including a brown allotropic form and a dark crystalline. The structure is diamond-like.
Which Element Does This Symbol Represent?
Phosphorus
The correct answer is “Phosphorus”.
Did you know…
A non-metallic chemical element, solid phosphorus has a molecular weight of 123.8952 atomic mass units and was discovered by Hennig Brand of Hamburg, Germany in 1674. Mr. Hennig prepared the element from urine. Later, in 1770, K. W. Scheele and J. G. Gahn discovered phosphoric acid in bone ash.
Which Element Does This Symbol Represent?
Sulfur
The correct answer is “Sulfur”.
Did you know…
Sulfur was known to ancient people. It is referred to in the Bible as “brimstone”. It was recognized as an element by A. L. Lavoisier in 1777. Solid sulfur is active in three forms. Brittle and yellow in color with no taste and insoluble in water, two of the forms are crystalline and one, “rhombic sulfur”, which is a crystalline structure orthorhombic in make-up. Liquid sulfur’s viscosity rises as it is heated. Long polymeric chains of molecules of sulfur make up this element.
What Element Does This Symbol Represent?
Chlorine
The correct answer is “Chlorine”.
Did you know…
A gaseous chemical element, this greenish gas is poisonous. It produces a suffocating odor that is approximately two and a half times more dense than air. Chlorine is one of the highest chemically active elements, with fluorine being the only one above it. Water soluble, the element will burn if it is ignited in a hydrogenated environment. When mixed with hydrogen, it can create explosive combinations.
What Element Does This Symbol Represent?
Argon
The correct answer is “Argon”.
Did you know…
Argon is a colorless, odorless and tasteless gas that is in air and in some volcanic gases. It is in the group of inert gases in group 18 of the periodic table. Argon is developed by fractional distillation of liquid air and has been a substitute for the use of nitrogen in common household light bulbs. It is incorporated with neon in the neon signs and the two produce a greenish glow. Argon is also used in arc welding as a protective atmosphere during the welding process.
What Element is This Symbol Representing?
Potassium
The correct answer is “Potassium”.
Did you know…
Potassium is in the category of alkali metals in group 1 of the periodic table. This limited-use element is extremely reactive and reacts violently with water. The combination of the two produces potassium hydroxide, or KOH, which releases hydrogen and can ignite. It reacts with many organic compounds as well.
Since potassium has such a close resemblance to sodium, it is not used as often due to the cheaper cost of sodium. Potassium compounds are still used in various industries, however, such as in soap and glass manufacturing.
Which Element is This?
Calcium
The correct answer is “Calcium”.
Did you know…
Calcium is a metallic chemical element that is silver to white in color and is somewhat of a soft metal with a cubic crystalline structure. Calcium is an alkaline-earth metal and falls into the category with the rest of these types of elements in group 2 of the periodic table. When it is exposed to air, it burns with a bright yellowish to reddish flame, forming nitride. Calcium will react to water. When combined with other elements such as oxygen, carbon, hydrogen, chlorine, fluorine, arsenic, phosphorus, and sulfur, it will form many new compounds.
The First Twenty Elements
These flash cards represent the first twenty elements of the element chart. Going through the symbols several times can assist you in the memorization of them. The elements are set up in various groups within the chart, according to chemical make-up. The number in the right hand corner is the number in which the element falls into the chart. For instance, Calcium is number 20 on the table, thereby displaying the number in the upper right hand corner. The “Ca” is the element itself and the symbol used in the discussion of calcium. The number on the bottom represents the electron arrangement that makes up the element.
References
All individual element symbols are by the author, “Me”; under CC BY-SA 2.5 license :
All data listed about chemical elements comes from “Interactive Periodic Table ”, Info Please Encyclopedia, Health & Science